Back to Publications
Zhigang Sun, Dequan Yu, Wenbo Xie, Jiayi Hou, Richard Dawes, Hua Guo, Kinetic isotope effect of the 16O + 36O2 and 18O + 32O2 isotope exchange reactions: Dominant role of reactive resonances revealed by an accurate time-dependent quantum wavepacket study, J. Chem. Phys. 142, 174312 (2015).
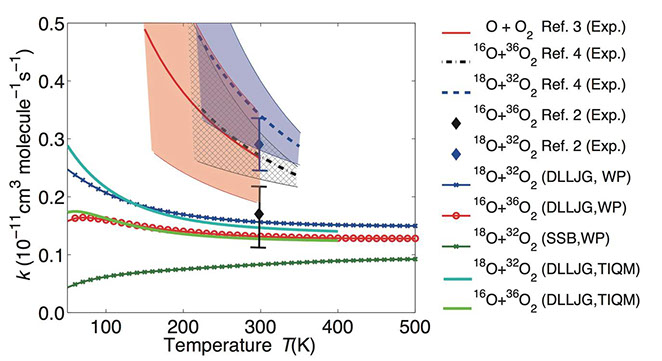
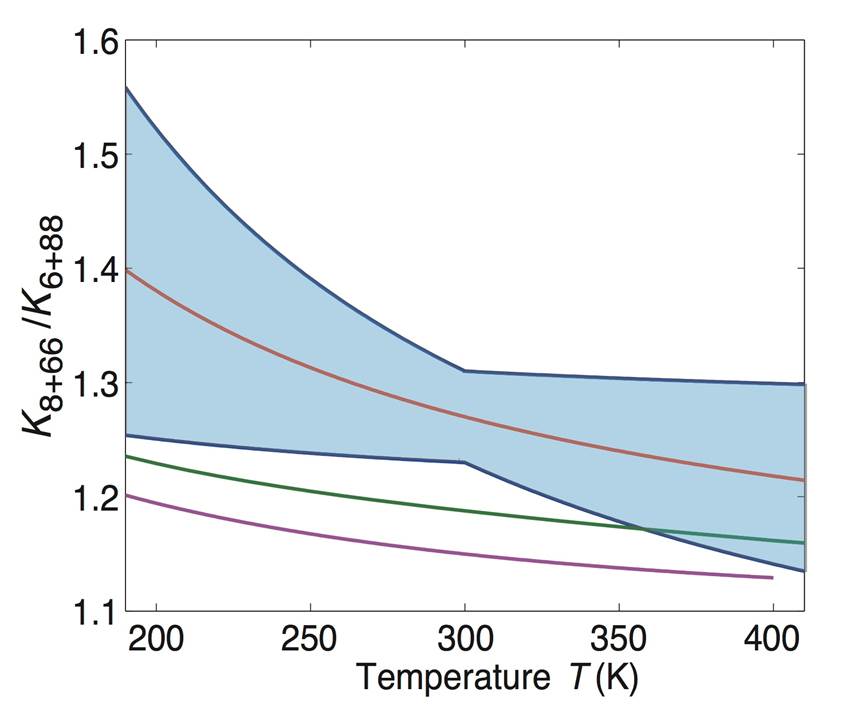
ABSTRACT: The O+O2 isotope exchange reactions play an important role in determining the oxygen isotopic composition of a number of trace gases in the atmosphere, and their temperature dependence and kinetic isotope effects (KIEs) provide important constraints on our understanding of the origin and mechanism of these and other unusual oxygen KIEs important in the atmosphere. This work reports a quantum dynamics study of the title reactions on the newly constructed Dawes-Lolur-Li-Jiang-Guo (DLLJG) potential energy surface (PES). The thermal reaction rate coefficients of both the 18O+32O2 and 16O+36O2 reactions obtained using the DLLJG PES exhibit a clear negative temperature dependence, in sharp contrast with the positive temperature dependence obtained using the earlier modified Siebert-Schinke-Bittererova (mSSB) PES. In addition, the calculated KIE shows an improved agreement with the experiment. These results strongly support the absence of the “reef” structure in the entrance/exit channels of the DLLJG PES, which is present in the mSSB PES. The quantum dynamics results on both PESs attribute the marked KIE to strong near-threshold reactive resonances, presumably stemming from the mass differences and/or zero point energy difference between the diatomic reactant and product. The accurate characterization of the reactivity for these near-thermoneutral reactions immediately above the reaction threshold is important for correct characterization of the thermal reaction rate coefficients.