Haber Process an Example of Keq
The Haber Process is the formation of Ammonia (NH3) from
nitrogen and hydrogen in their gaseous forms (N2 and H2 respectively). This is given by the equation:
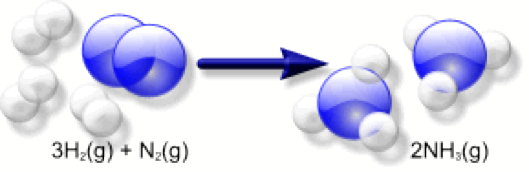
3H2 (g) + N2(g) → 2NH3(g)
This
reaction is exothermic, and is speeded up by an iron catalyst.