The position of an
equilibrium is affected by many things.
This means that the relative concentrations of reactant and products can differ. Although the rate
of the forward reaction is the same as the
rate of the reverse reaction, you can still
start off with more ammonia. However, equilibria are affected by concentration.
Haber Process an Example of Keq
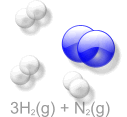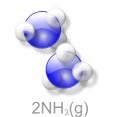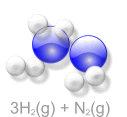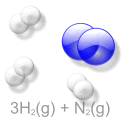
If there is more
ammonia, the equilibrium will
shift to try and make things
equal again. This means the reverse
reaction will speed up a bit, until the
concentrations are where they want to be, and
then it will return to a constant
dynamic equilibrium - forward and reverse reactions being the same.