Chromatography is based on a physical equilibrium
that results when a solute is transferred between the mobile and a stationary phase. A
A
A
A
A
A
A
A
A
A
A
A
K =
distribution
coefficient or
partition ratio
K
=
C
S
C
M
Where CS is the molar concentration
of the solute in the
stationary phase and CM is the molar concentration in the mobile phase.
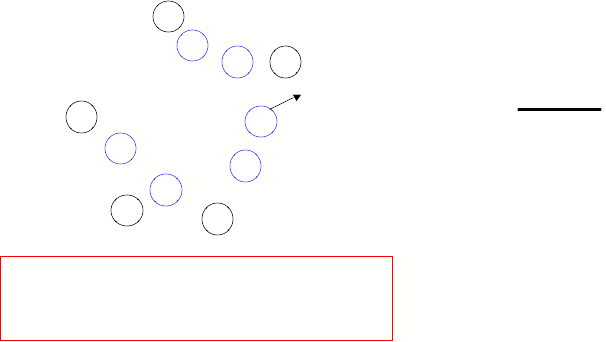
Cross Section of
Equilibrium in a column.
“A” are adsorbed to the
stationary phase.
“A” are traveling in the
mobile phase.