
pH 0
[H3O+] 100
[OH] 10-14
pOH 14
1
10-1
10-13
13
2
10-2
10-12
12
3
10-3
10-11
11
4
10-4
10-10
10
5
10-5
10-9
9
6
10-6
10-8
8
7
10-7
10-7
7
8
10-8
10-6
6
9
10-9
10-5
5
10
10-10
10-4
4
11
10-11
10-3
3
12
10-12
10-2
2
13
10-13
10-1
1
pH = -log [H3O+]
pOH = -log[OH-]
pH + pOH = 14
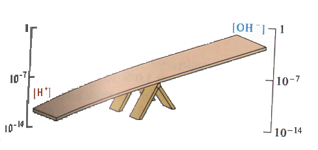
What is pH?
14
10-14
100
0