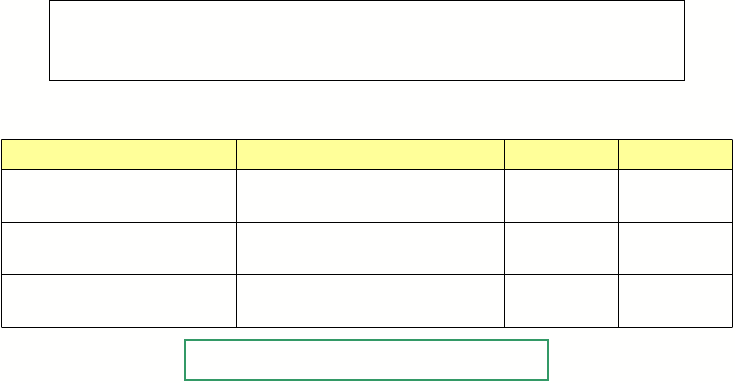
Formula masses and percent
composition
of three theoretical compounds of Mg
and O
Empirical
Formula Example 2
2nd Step: Formula Masses and their Percent Composition.
Mg = 24.31 g/mole
O = 15.999 g/mole
15.999/ 64.619
2 x 24.31/ 64.619
64.619
2(24.31) + 15.999 =
Mg2O
2 x 15.999/ 56.308
24.31/ 56.308
56.308
24.31 + 2(15.999) =
MgO2
15.999/ 40.309
24.31/ 40.309
40.309
24.31+15.999
=
MgO
%O
%Mg
Formula
Weight
Formula
of Oxide MgxOy
Mg = 60.3% and O =
39.7%