Reaction:
1 NaHCO3(aq) + CH3CO2H(aq) ®
_____ + 1 CO2(g) + ______
Expected mole of CO2(g) : ___________ moles
Expected volume of gas at laboratory T & P: _____L
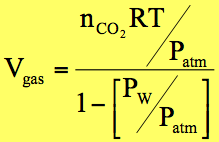
Lab temp. (K)
0.08206 L.atm/mole.K
Lab pressure (atm)

Partial pressure of water vapor (atm) (calculated in
Part 1.)
calculate