A Calorimeter
may be used to determine the Heat Capacity, Cs, of a material by measuring the temperature change when
a known mass of the
material at a higher temperature is placed in a known mass of water, usually at room temperature, and the system is allowed to
reach a final intermediate temperature.
Heat lost by hot object = Heat gained by cold water
Csmaterial(mass)material(Tfinal-Tinitial)material = Cswater(mass)water(Tfinal-Tinitial)water
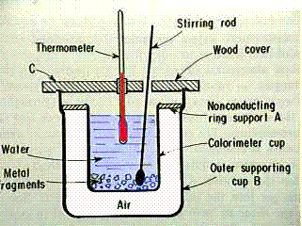

Note: The heat
capacity is related to the atomic
mass and the intermolecular forces
in the material.
Calorimetry